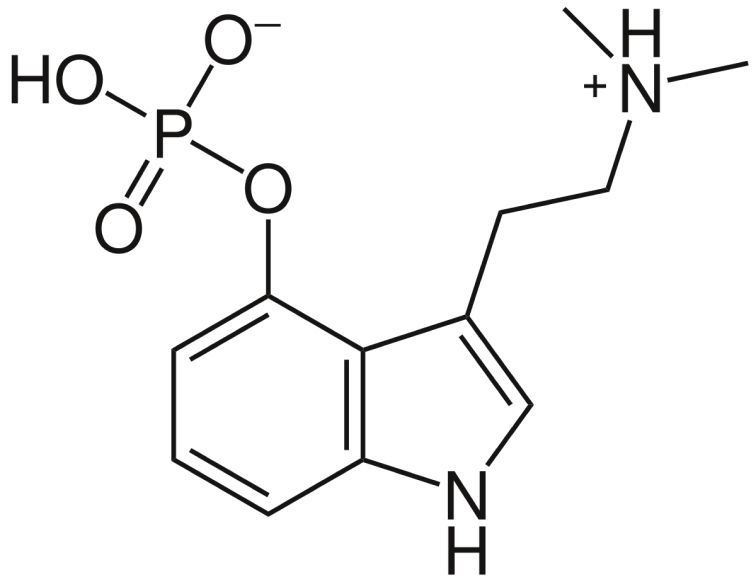
Since Psilocin begins to degrade almost immediately when exposed to oxygen, we have developed a delivery media that protects the active from coming into contact with air.
Also, since psilocybin potency diminishes upon exposure to water and UV light, our novel carriers and encapsulation process utilize both anhydrous carrier liquids and UVA&B resistant gelatins for our precisely dosed Soft gel capsules.
As legalized medicinal use of psilocin, psilocybin and other naturally derived tryptamines is a new frontier, this leaves open a great deal of opportunity to seize the market on new FDA approved formulations.
In addition to novel formulation development, there is also the opportunity for IP development for the mechanized manufacturing of these new medicines that incorporate our Licensed delivery systems and mechanisms.